Edelife XLHED Clinical Trial
Now Enrolling Pregnant Women
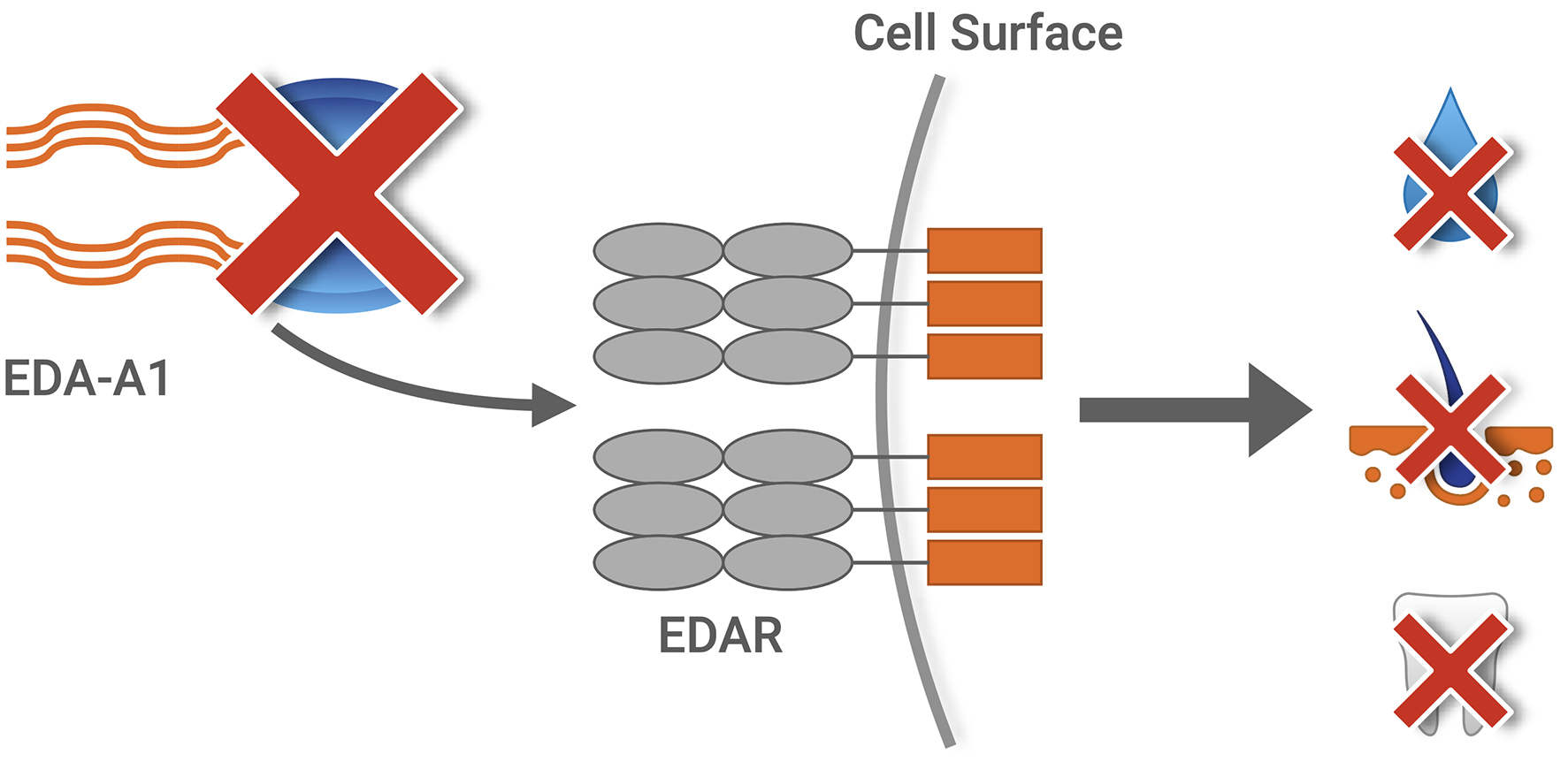
X-Linked Hypohidrotic Ectodermal Dysplasia (XLHED), the most common form of ectodermal dysplasia, is caused by a broad spectrum of mutations in the ectodysplasin A1 (EDA-A1) gene. The main symptoms of XLHED are hypohidrosis (or anhidrosis), oligodontia (or anodontia) and hypotrichosis. Current treatment options are limited to managing disease symptoms and complications. Effective corrective treatment for XLHED remains a high unmet medical need.
EDELIFE is a clinical research study evaluating the safety profile and potential efficacy of an investigational product in pregnant women who are carrying a boy diagnosed with X-linked hypohidrotic ectodermal dysplasia (XLHED). The investigational product has not been approved by regulatory authorities.
Investigational Protein Intended to Replace EDA-A1 in utero
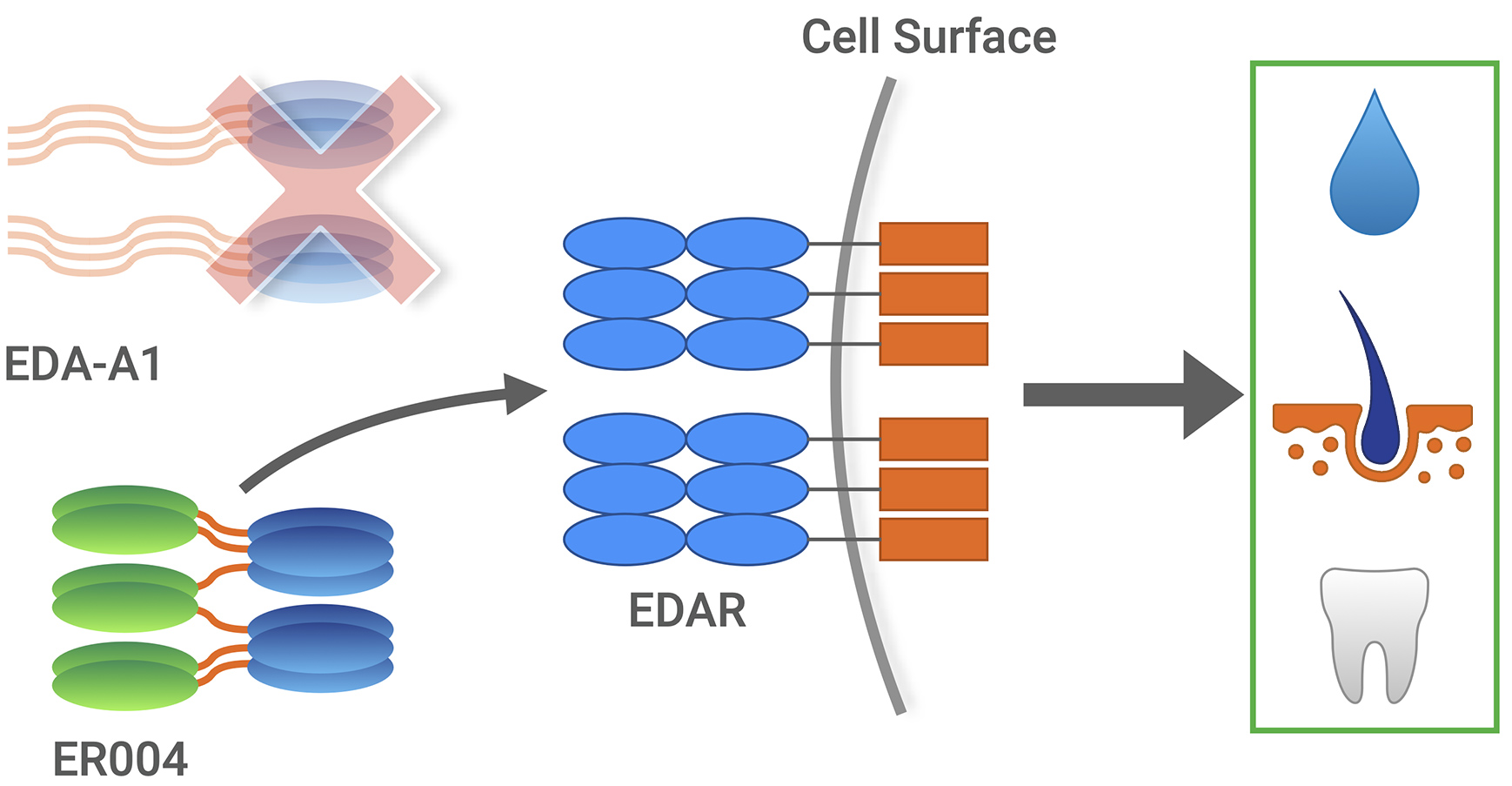
The Edelife investigational treatment, ER004, consists of a signaling protein replacement molecule designed for specific, high affinity binding to the endogenous receptor, intended to replace missing EDA-A1 protein in utero for patients with XLHED.
Administration at several specific times in pregnancy is essential, as ER004 impacts development of ectodermal precursors of sweat glands, teeth, etc.
EDA-A1 is missing in XLHED
ER004 as an intended replacement for EDA-A1
Prior Use of ER004 in XLHED Patients
Prior Use of ER004 in XLHED Patients
- Results from three XLHED-affected boys treated in utero with ER004 were first reported in the New England Journal of Medicine in 2018.1
- Long-term results (N=6 patients, follow-up range 2 to 6 years) were recently published in the International Journal of Molecular Sciences in 2023.2
- The aim of the Edelife trial is to evaluate the safety and efficacy of ER004 administered prenatally.

NEXT STEPS: EDELIFE STUDY ENROLLMENT
References: 1. Schneider H, Faschingbauer F, Schuepbach-Mallepell S, Körber I, Wohlfart S, Dick A, Wahlbuhl M, Kowalczyk-Quintas C, Vigolo M, Kirby N, Tannert C, Rompel O, Rascher W, Beckmann MW, Schneider P. Prenatal Correction of X-Linked Hypohidrotic Ectodermal Dysplasia. N Engl J Med. 2018 Apr 26;378(17):1604-1610. 2. Schneider, H.; Schweikl, C.; Faschingbauer, F.; Hadj-Rabia, S.; Schneider, P. A Causal Treatment for X-Linked Hypohidrotic Ectodermal Dysplasia: Long-Term Results of Short-Term Perinatal Ectodysplasin A1 Replacement. Int. J. Mol. Sci. 2023, 24,7155


